REGENER-EYES®
Regener-Eyes® Government Mission Statement:
Our mission is to develop innovative, pharmacologic solutions that empower physicians and clinicians to provide the best in care to our military service members and veterans.
Regener-Eyes® Government Mission Objective:
Our mission is to help our veterans and military service members gain easy and affordable access to life-changing medicines. We accomplish this by making Regener-Eyes® available to the VA, DOD, FEMA, and other government agencies through special contracting vehicles, the VANF and Tricare formularies, our pharmaceutical and SDVOSB Distributor partners, and through other federal procurement pathways.
Regener-Eyes® is Proudly:
Made and Manufactured in the USA, FDA Registered and Compliant
Trade Agreement Act Compliant
Berry Amendment Compliant
Buy American Act Compliant
Commonly Used and Recommended Within The VA Healthcare Network, and The Military Health System.
Regener-Eyes® Safety:
Regener-Eyes® offers a safe and effective solution as a more affordable option.
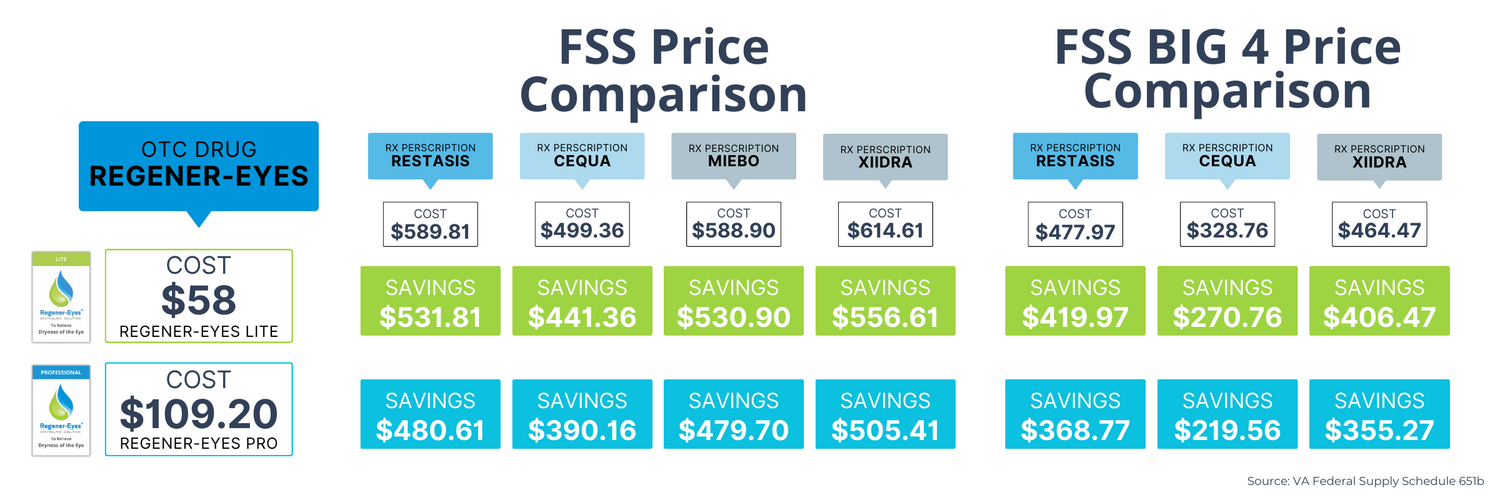
POTENTIAL COST SAVINGS
EXPLORING REGENER-EYES MECHANISM OF ACTION
Regener-Eyes® Tonicity Solution™ employs patented technology to combat dry eye-triggering hyperosmolarity

Download Capabilities PDF
GOVERNMENT POC
Corporate Data
CAGE: 79YW0 | UEI: N2ZTM3FCKHN5
Work Area: Nationwide
Gov. Bus. POC: Valentino Pine, Director of Government Relations
Phone: (813) 901-4064
E-Mail: Valentino@RegenerativePlant.org
Website: www.RegenerEyes.com
Socio-Economic Status: Small Business Enterprise (SBE)
Primary NAICS: 325412: Pharmaceutical Preparation Manufacturing
Distributor Contact Info
CAGE: 1TAP7 | UEI: VSSNJEFMPLB3VA FSS 651b
Contract #: 36F79719D0161
Gov. Bus. POC: Samir Patel, President
Gov. POC Email: Samir@MarlexPharm.com
Phone: (888) 582-1953
Fax: (302) 898-1609
E-Mail: CustomerService@MarlexPharm.com
Website: www.MarlexPharm.com

NAICS & PSC Codes
NAICS Codes:
Primary 325412: Pharmaceutical Preparation Manufacturing
339115: Ophthalmic Goods Manufacturing
339113: Surgical Appliance & Supplies Manufacturing
541690: Other Scientific & Technical Consulting Services
541714: Research & Development in Biotechnology
541990: All Other Professional, Scientific & Technical Services
541715: R&D, Physical, Engineering & Life Sciences
(Non-Defense Applications)
PSC Codes:
6505: Drugs & Biologicals
6540: Ophthalmic Instruments, Equipment & Supplies
NDC:
82305-0003-02 Regener-Eyes Pro
82305-0006-02 Regener-Eyes Lite
Mckesson Item Number:
2823243 Regener-Eyes Pro
2823359 Regener-Eyes Lite

CORE COMPETENCIES
Regener-Eyes® Ophthalmic Solution helps increase lubrication and hydration of the corneal surface. This helps to relieve dryness of the eye.
Regener-Eyes® Ophthalmic Solution has an excellent safety profile.
Regener-Eyes® is non-steroidal. It helps to protect and relieve dryness of the eye.
Each batch of Regener-Eyes® is tested to ensure quality, sterility, and safety.
Regener-Eyes® Ophthalmic Solution, can be stored at room temperature until use. Once opened, each bottle can be stored at room temperature and used within 90 days.
Regener-Eyes® Ophthalmic Solution, has a shelf life of two (2) years when stored at room temperature.
PAST PERFORMANCE
-
Department of Veterans Affairs
Strategic Acquisition Center Contract
Contract #: 36C106
Date(s): 2018-Present | Type: IDC
Details: Drugs & BiologicsDepartment of Veterans Affairs
Date(s): 2023-Present | Value: $40,000
Details: Provided Amniotic Membrane and Regener-Eyes LITE to Network Contracting Office 19 & 16 through the VANF.Department of Defense
Date(s): 2023-Present | Role: Subcontractor
Details: Provided Regener-Eyes LITE through distributor Marlex Pharmaceuticals to DoD prime vendor: McKesson
JAMES LEWIS, MD
Board Certified Ophthalmologist, Doctor James S. Lewis, Director of Cataract, LASIK and Corneal Surgery (Pennsylvania College of Optometry, Salus University) shares his experience using Regener-Eyes® in his clinical practice.
DÁMARIS RAYMONDI, OD, FAAO
Dr. Damaris Raymondi is the New York State Young Optometrist of the Year 2023. She received her Doctor of Optometry degree from the SUNY College of Optometry in 2015. She splits her time between NYC Health + Hospitals/Metropolitan and her practice Sur Eye Care. Her focus is on pediatrics and ocular disease.
Optometry Times: Helping Dry Eye Associated with Sjögren's Syndrome: A Case Study By Dámaris Raymondi, OD, FAAO

Read White Paper
The Molecular Mechanisms Responsible for Tear Hyperosmolarity-Induced Pathological Changes in the Eyes of Dry Eye Disease Patients
This research delves into the molecular mechanisms underlying Tear Hyperosmolarity (THO) in Dry Eye Disease (DED). Driven by a collaborative effort between scientists and healthcare experts, spearheaded by Dr. C. Randall Harrell, it introduces a groundbreaking approach to treating dry eye by elucidating the fundamental causes of the condition. The paper highlights THO as a key player in the onset and progression of DED, contributing to discomfort, inflammation, and eye damage. The findings propose innovative solutions, such as personalized eye drops, aimed at restoring tear composition. Additionally, the research advocates for exploring hypo-osmotic eye drops and NF-κB inhibitors to enhance the effectiveness of DED treatment.

READ WHITE PAPER